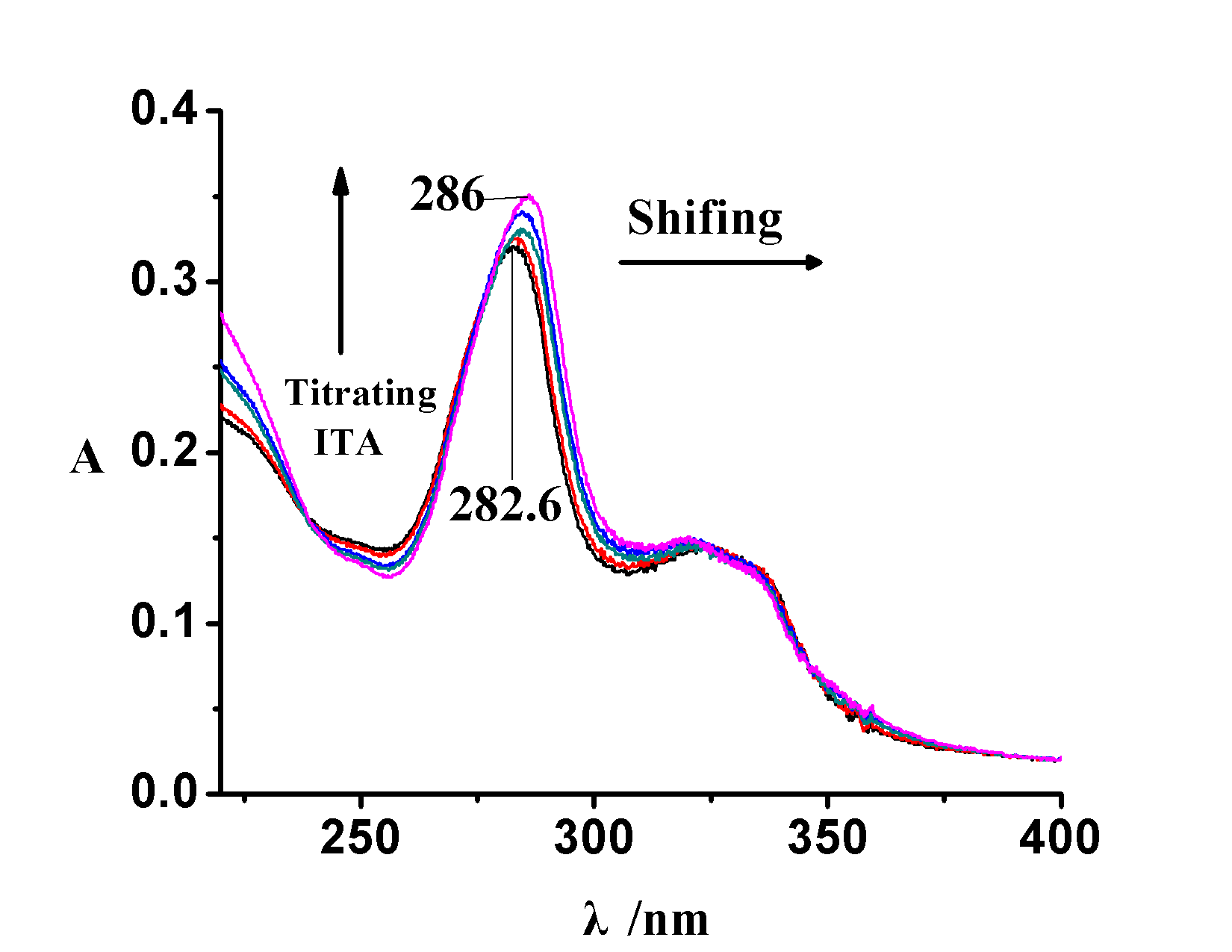
In this Chemistry Magazine,you have chemistry related news about a molecularly imprinted polymers (MIP) for sarafloxacin was prepared by the use of itaconic acid as the functional monomer and ethylene glycol dimethacrylate as the crosslinking monomer. But we will also show you the funny side of chemistry, as we earlier discussed the games we stumbled on. Why don’t you take a free test at the casino laboratory by discovering the best casino bonus chips at freecasinogameschips.com? You will find many games that will satisfy your scientific potential. Not let’s go back to the real topic, shall we? To improve selectivity, cobalt(II) ions was used to form metal-template complex, which provided the specific binding sites similar to metal-chelate antibody. The influences of different anions and cations on the recognition performance of the MIP were investigated in protic solvent. Due to the specific structure of the complex of cobalt(II)-sarafloxacin, it increased …
Effects of cation-anion on recognition characteristics of sarafloxacin -Co(II) complex imprinted polymer in aqueous media the other tested compoun