Liu Hanxing, Wang Xian, Hu Weida , Guo Liling , Ouyang Shixi
(State Key Laboratory of Advanced Technology for Materials Synthesis and Processing, Wuhan University of Technology, Wuhan 430070, China)
Received Mar. 8, 2004; Supported by Key Program of the National Nature Science Foundation of China (No.90206047)
Abstract Organic-inorganic layered perovskite-type compounds (C4H9NH3)2CuX4 (X=Cl, Br) were prepared from solutions in an air atmosphere. X-ray diffraction, Scanning electron microscopy (SEM), FTIR spectroscopy and thermal analysis (TG and DSC) were used to characterize the obtained powders. From the SEM pictures, both of (C4H9NH3)2CuCl4 and (C4H9NH3)2CuBr4 took on obvious sheet-like microstructure, as well as their XRD profiles demonstrated the crystals growth were highly oriented. The FTIR spectrum showed that the characteristic peaks of (C4H9NH3)2CuCl4 are shifted to higher frequency due to protonation of C4H9NH2. The thermal stability of (C4H9NH3)2CuBr4 was inferior to (C4H9NH3)2CuCl4 due to its higher Jahn-Teller distortion of inorganic sheets.
Keywords layered perovskite-type, characterization, microstructure, thermal stability
-
INTRODUCTION
Layered organic-inorganic perovskite-type materials have been studied intensively due to their potential for unique electrical, magnetic, and optical properties. In these system, organic and inorganic components combined into a molecular material. Organic components provide virtually unlimited flexibility to choose molecules of varying length, width, polarizability, and degree of saturation or polymerization, as well as offering high luminescent properties, large polarizability, plastic mechanical properties, ease of processing, and structure diversity. Inorganic components, on the other hand, offer the potential for a wide range of electronic properties (enabling the design of insulators, semiconductors, and metals), magnetic and dielectric transitions, substantial mechanical hardness and thermal stability[1-5]. The (RNH3)2MX4(M=divalent metal, X=halogen) members of the perovskite-type family have been synthesized from solution and have generated considerable interest as self-assembling multi-quantum-well structures, where inorganic layers of two-dimensional network of corner-sharing octahedral metal halide MX6 and organic layer of molecular ammonium cation RNH3 are alternately piled up, R can be substituted by alkyl group, chromophore, etc [6,7]. Due to the low dimensionality of the inorganic sheets, the exciton has a large binding energy of several millielectron volts, which enables strong photoluminescence even at room temperature sufficient to offer potential applications in emitter materials in electroluminescent devices[8-12]. In this paper, we report the preparation of (C4H9NH3)2CuX4 (X=Cl, Br) by a solution method in an air atmosphere and characterize their layered perovskite-type structures, furthermore test their thermal stabilities. The reason why we choose the titled systems lies in two facts. Firstly, numerous papers have reported there were phase transitions in the layered (RNH3)2CuCl4 systems and caused which properties changing at different temperatures, the prepared new crystals of butylammonium tetrahalocuprate (C4H9NH3)2CuX4 should be considered as two-dimensional materials according to the butyl chain length, in the future work, we would also study their electronic and magnetic properties in comparison with the analogous systems (RNH3)2CuCl4 reported in the publication papers [8,11,13]. Secondly, the differences of crystal structure and thermal stability between the titled materials can help us to conclude the effects on the compound due to different halogen. -
EXPERIMENTAL
2.1 Sample preparation
Crystal of the title compounds were grown from slowly cooled aqueous HCl acid solution in the air. The compound (C4H9NH3)2CuCl4was synthesized by first dissolving 6.8192g (0.04mol) of CuCl2·2H2O in 25ml aqueous HCl (37%wt) at 70ºCin a water bath, and 7.9072ml (0.08mol) of C4H9NH2 in 6.6246ml (0.08mol) aqueous HCl (37%wt) to protonation. Upon dropwise addition of the organic ammonium solution into the CuCl2 solution, drastically reaction began and green precipitate immediately formed. The solution was allowed to cool and filtered at decompression. Then the green powders turned to yellow and was dried in vacuum dry box at 70ºC.
Crystals of (C4H9NH3)2CuBr4 was prepared by a method analogous to the X=Cl. Briefly, 6.7011g (0.03mol) of CuBr2 was dissolved in 30ml aqueous HBr (40%wt) at 70ºC in a conical flask, and 5.9303ml (0.06mol) of C4H9NH2 in 8.7946ml (0.06mol) aqueous HBr (40%wt) in a separate beaker and then added to CuBr2 solution. After cooling the solution, the purple crystals formed was filtered and dried in vacuum at 70ºC.
Element analysis for the two products was taken by Elementar Analysensysteme GmbH VarioEL III, the found and [theoretical] results are illustrated in Table 1. It can be seen the found value is close to the proposed C8H24N2CuX4 formula.Table 1. The C, H, N content of (C4H9NH3)2CuX4 (X=Cl, Br)
| Sample | (C4H9NH3)2CuCl4 | (C4H9NH3)2CuBr4 | |
| Content (%) | C | 27.37 [27.17] | 18.06 [18.08] |
| H | 6.520 [6.84] | 4.476 [4.55] | |
| N | 7.779 [7.92] | 4.975 [5.27] | |
2.2 Characterization
The crystallinity and orientation of the powder samples were determined by X-ray diffraction. X-ray diffraction (XRD) measurements recorded with a 2q ranging from 5 to 70º were performed on a Japan D/MAX-YB with a CuKa target at 40 kV and 50 mA. Microstructure of powders were analyzed by Japan JSM-5610 scanning electron microscopy (SEM). The infrared transmission spectrums for butylamine and (C4H9NH3)2CuCl4 were taken by NICOLET 60-SXB FTIR spectroscopy. Simultaneous thermogravimetric apparatus (TG) and differential scanning calorimetry (DSC) were performed on the two samples in an argon atmosphere.
-
RESULTS AND DISCUSSION
3.1 X-ray diffraction
Fig.1 shows X-ray diffraction profiles of powders of (a) (C4H9NH3)2CuCl4and (b) (C4H9NH3)2CuBr4. It can be seen from Fig.1 both of the two examples have series of (00l) diffractions, with nearly no extra undexed Bragg reflections, this result shows that the two examples having formed are both highly oriented in the c-axis direction. For (C4H9NH3)2CuCl4, the interlayer d-spacing value calculated from the XRD is about 15.4058Å, however, this value of (C4H9NH3)2CuBr4is 14.7941Å, marked less than which of (C4H9NH3)2CuCl4. We find that the unit-cell volume of (C4H9NH3)2CuBr4 is accordingly decreased because the peaks of which diffraction occurred at higher angles, although the radius of Cl–(181pm) is less than that of Br–(195pm). We assume that this could be attributed to the deformation of the inorganic layers caused by Jahn-Teller distortion of CuX6 octahedral. As for the color of the (C4H9NH3)2CuCl4 powders turned to yellow from green, it should be due to the structure transition, each octahedron is lengthened along the Jahn-Teller z axis, which lies in CuCl plane and has nearly orthogonal relations with the neighbors[8,13].
Fig.1 XRD profiles for powder of (C4H9NH3)2CuCl4 (a) and (C4H9NH3)2CuBr4 (b)
3.2 Scanning electron microscopy
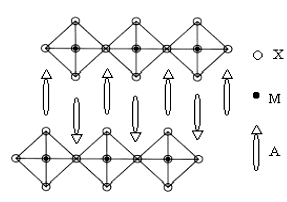
Fig.2 shows SEM pictures of powders of (a) (C4H9NH3)2CuCl4 and (b) (C4H9NH3)2CuBr4.It can be seen from Fig.2 that the two samples take on obvious sheet-like microstructures. We have known that organic-inorganic layered perovskite-type assembling materials A2MX4 derive from three-dimensional perovskite structure, where M is a divalent metal, X is a halide and A can be a wide range of organic amine. As shown in Fig.3, at the molecular level, the 2-D inorganic MX42- layer and organic ammonium layer are alternately stacked along c-axis [14,15]. We deemed that the two samples’ sheet-like microstructures and highly orientation growth, as well as confirmed by their XRD profiles, are mainly due to the high oriented 2-D layered molecular structure of (C4H9NH3)2CuCl4 and (C4H9NH3)2CuBr4.
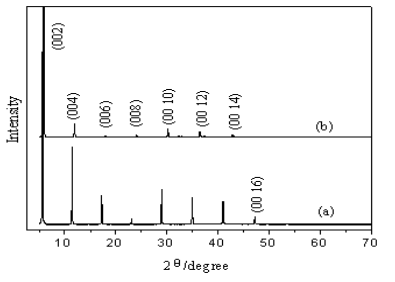
Fig.2 SEM micrographs of sample (C4H9NH3)2CuCl4 (a) and (C4H9NH3)2CuBr4 (b)
3.3 Infrared transmission analysis
Fig.4 shows FTIR spectrum of (a) C4H9NH2 and (b) (C4H9NH3)2CuCl4. It can be seen from Fig.4 that the intensity and shape of the characteristic peaks exhibit obvious differences between the reactant and precipitate. The characteristic peaks in the compound include approximately 3439 cm-1, 3168 cm-1, 3131 cm-1, 1575 cm-1 and 1487 cm-1, which are broadened and intensified in comparison to the free amine, moreover, the peaks are shifted to higher frequency (as well as in the (C4H9NH3)2CuBr4). These variations presumably ascribe to protonation of C4H9NH2 [14].
Fig.3 Schematic structure of layered perovskite-type A2MX4
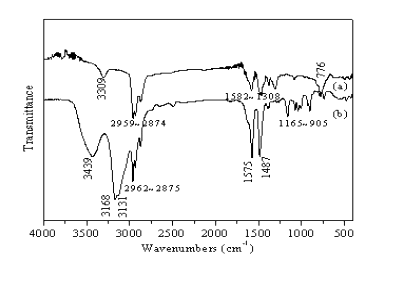
Fig.4 FTIR spectrum for C4H9NH2 (a) and (C4H9NH3)2CuCl4 (b)
3.4 Thermal analysis
The results of TG-DSC shows the decomposition of (C4H9NH3)2CuCl4 occurs at 261.6ºC, and (C4H9NH3)2CuBr4 at 197.0ºC respectively. The thermal stability of (C4H9NH3)2CuCl4 is obviously higher than that of (C4H9NH3)2CuBr4. According to the weight loss curve, we assume that gradual heating of (C4H9NH3)2CuX4(X=Cl, Br) compounds leads to the two step dissociation of the organic component from the structure:
(C4H9NH3)2CuX4 (s)→C4H9NH3X (g) + C4H9NH3CuX3 (s) →2C4H9NH3X (g) + CuX2(s) [15].
Because of the Jahn-Teller effect on [CuX6] inorganic sheets in (C4H9NH3)2CuX4, the N―H…X hydrogen binding is weakened and the electrostatic repulsion is increased between the halide ion in the CuX42- anions [13]. We consider the dissociation of (C4H9NH3)2CuBr4 to C4H9NH3Br is easier than C4H9NH3Cl from (C4H9NH3)2CuCl4, presumably due to higher deformation of which inorganic sheets.
-
CONCLUSIONS
In air ambient, the layered perovskite-type powders of (C4H9NH3)2CuX4 (X=Cl, Br) can be acquired using a solution technique. The obtained crystals had high orientation and their microstructures took on obvious sheet-like. The FTIR spectrum show us the amine is protonation in the compound, which is in agreement with theoretical assumption. The crystal structure and thermal properties of (C4H9NH3)2CuX4 (X=Cl, Br) has some differences due to the sort of halide anion, as we note that (C4H9NH3)2CuBr4 has less d-spacing value and lower decomposition point than (C4H9NH3)2CuCl4. For the compounds (C4H9NH3)2CuX4 (X=Cl, Br), there should be interesting electronic and magnetic properties ascribed to their characteristic structures as other systems having been reported, and further study would be continued.
REFERENCES
[1] David B M, Chondroudis K, Kagan C R. IBM J. RES.& DEV., 2001, 45 (1): 29.
[2] Masanao Era et al. Thin Solid Films, 2001, 393: 24.
[3] David B M. Chem. Mater., 1996, 8: 79.
[4] Fujita T, Nakashima H, Hirasawa M et al. Journal of Luminescence, 2000, 87: 847.
[5] Etienne Wortham et al. Physical B, 2002, 318: 387.
[6] Braun M, Tuffentsammer W, Wachtel H et al. Chemical Physics Letters, 1999, 307: 373.
[7] Braun M, Tuffentsammer W, Wachtel H et al. Chemical Physics Letters, 1999, 303: 157.
[8] Salim Haddad, Roger D. Willett. Inorg. Chem., 2001, 40: 2457.
[9] Shigeki Kashiwamura, Nobuaki Kitazawa. Synthetic Metals, 1998, 96: 133.
[10] Papavassiliou G C, Mousdis G A, Koutselas I B. Synthetic Metals, 2001, 121: 1339.
[11] Eji Shikoh, Yasuo Ando, Masanao Era et al. Journal of Magnetism and Magnetic Materials, 2001, 226-230: 2021.
[12] Kazuhiro Sumioka, Hiroyuki Nagahama, Tetsuo Tsutsui. Applied Physics Letters, 2001, 78 (10): 1328.
[13] Taketoshi Sekine, Tsunehisa Okuno, Kunio Awaga. Chemical Physics Letters, 1996, 249: 201.
[14] David B M, K. Liang. Journal of solid state chemistry 134, 1997: 376.
[15] David B M et al. Chem. Mater., 1999, 11:542.
