Abstract Kinetics’ and Thermodynamics of The Adsorption of of Ni 2+ Onto Chitosan-Aluminum Composite Material’s WAS Studied oxide. The Under Experimental Conditions, s Indicated Kinetic Adsorption of that of Ni 2+ Onto Aluminum oxide-Chitosan Composite Material’s Conforms to pseudo-SECOND-Order Equation And the apparent adsorption activation energy is 28.62 kJ/mol.In the experimental temperature, Chemistry Magazine Online and thermodynamic studies indicated that adsorption of Ni 2+ onto chitosan-aluminium oxide composite material conforms to Langmuir adsorption isotherm equation. Enthalpy of adsorption ( D H) value is 15.93 kJ/mol and entropy of adsorption( D S)value is 95.43J/(mol· K).Gibbs free energy ( DG) decreases with increasing temperature.
Keywords chitosan; composite material; adsorption;
Adsorption Kinetics and Thermodynamics of Ni 2+ on Chitosan-Aluminum Oxide Composites
MA Zhi-guang, ZHANG Fang, GENG Na, LIU Su-wen, LIU Pan
(College of Chemistry and Environmental Science, Hebei University, Baoding, Hebei 071002, China)
Abstract The kinetics and thermodynamics of Ni 2+ adsorption on chitosan – aluminum oxide composites were studied. The results show that the adsorption accords with the pseudo second-order kinetic equation and the apparent activation energy E a = 28.61 kJ/mol under the experimental conditions. The thermodynamic studies show that the adsorption conforms to the Langmuir isotherm equation at the experimental temperature and the adsorption enthalpy change D H q At 15.93 kJ/mol , the entropy change D S q =95.43 J/mol.K . The adsorption Gibs free energy D G q decreases with increasing temperature.
Key words chitosan composites adsorption kinetics thermodynamics
Nickel is the first type of pollutant that is strictly controlled in sewage discharge. Nickel-containing wastewater mainly comes from smelters and electroplating plants. After entering the water body, nickel cannot be degraded by microorganisms but can be enriched by aquatic organisms and enter the food chain to endanger humans. Adsorption is one of the main methods for removing Ni 2+ from industrial wastewater .
Chitosan is a product of deacetylation of natural polymer material chitin. There are a large number of hydroxyl and amino groups on the molecular chain, which can be used as adsorbents for the adsorption of heavy metal ions in wastewater [1] . However, the existence of hydrogen bonds between the chitosan molecular chains and the molecular chains affects the adsorption capacity, and poor acid resistance and mechanical strength need to be further improved. Xie Guangyong et al. [2] used chitosan and aluminum isopropoxide as raw materials to prepare chitosan-aluminum oxide composites by chemical bonding method. The adsorption performance of copper ions and mercury ions was greatly improved and stable. Sex has been improved. The adsorption kinetics and thermodynamic characteristics
of Ni 2+ in solution in chitosan-aluminum oxide composites were studied . It is helpful to investigate the regularity of adsorption of metal ions in aqueous solution by composites.
1 Experimental part
1.1 Instruments and reagents
UV-Vis spectrophotometer (WFZ800-D3A type, Beijing Second Optical Instrument Factory); water bath oscillator; electronic balance.
Chitosan (degree of deacetylation >90%, Zhejiang Jincai Biochemical Co., Ltd.); aluminum isopropoxide (analytical pure, Sinopharm Chemical Reagent Co., Ltd.); NiSO 4 ? 6H 2 O, toluene, ethanol, etc. pure.
1.2 Experimental method In the method described in
reference [2] , 100 mL of dry toluene and 3 g of aluminum isopropoxide were sequentially added to a nitrogen-protected 250 mL three-necked flask equipped with a reflux condenser, and magnetic stirring was performed at 50° C. for 30 min before adding 10 g. Chitosan was heated to 112° C. and refluxed for 5 h to stop the reaction. After filtration, the product was washed with anhydrous toluene, absolute ethanol, and distilled water 6 times in sequence. Finally, the product was placed in an oven at 80°C and dried to obtain a chitosan-aluminum oxide composite material.
Accurately weigh 0.5000±0.0002g of the composite into a 50ml Erlenmeyer flask, and accurately pipette 20ml of a nickel sulfate solution with a known concentration into a water bath shaker and shake at a set temperature (130r × min -1 ) Rapid filtration after a certain period of time. The filtrate was taken to determine the absorbance at 721 nm using an ultraviolet-visible spectrophotometer, and the residual Ni 2+ concentration was calculated according to a standard curve . The amount of adsorption is calculated according to equation (1):
Q = (C o -C) × V/m (1)
where Q is the amount of adsorption (mmol ×G -1 ); C O and C a solution of Ni 2+ concentration (mol and at the end of the initial adsorption × L -1 ); V is the volume of solution (mL); m is the mass of adsorbent (g).
2 Results and discussion
2.1 Kinetics of the adsorption of Ni 2+ on composites The kinetics of the adsorption of Ni 2+ on
composites at different times and temperatures are shown in Fig. 1 for a concentration of 0. 1200mol× L -1 Ni 2+ . As can be seen from Figure 1 , the adsorption of Ni 2+ by the composite material has better adsorption performance. This adsorption is basically in accordance with the three necessary steps for adsorption of the substance in the solution on the porous adsorbent [3,4] . At the beginning, the adsorption rate was faster, and it decreased regularly with the extension of time, and it tended to equilibrate after 360 minutes . This is because at the beginning, Ni 2+ is mainly adsorbed on the outer surface of the composite material and the adsorption is faster. With the progress of the adsorption process, the Ni 2+ concentration gradually decreases, and the adsorbate is inward along the pores of the composite material. Diffusion and diffusion resistance increase gradually, and the adsorption rate is mainly controlled by diffusion, which results in a slower adsorption rate. In the late stage of adsorption, adsorption occurs mainly on the inner surface of the adsorbent, and the concentration driving force is getting smaller and smaller, and adsorption has basically reached equilibrium. When the temperature rises, the on activated molecules percentage increases, so that the adsorption rate increases.
Figure 1 Chitosan at different temperatures
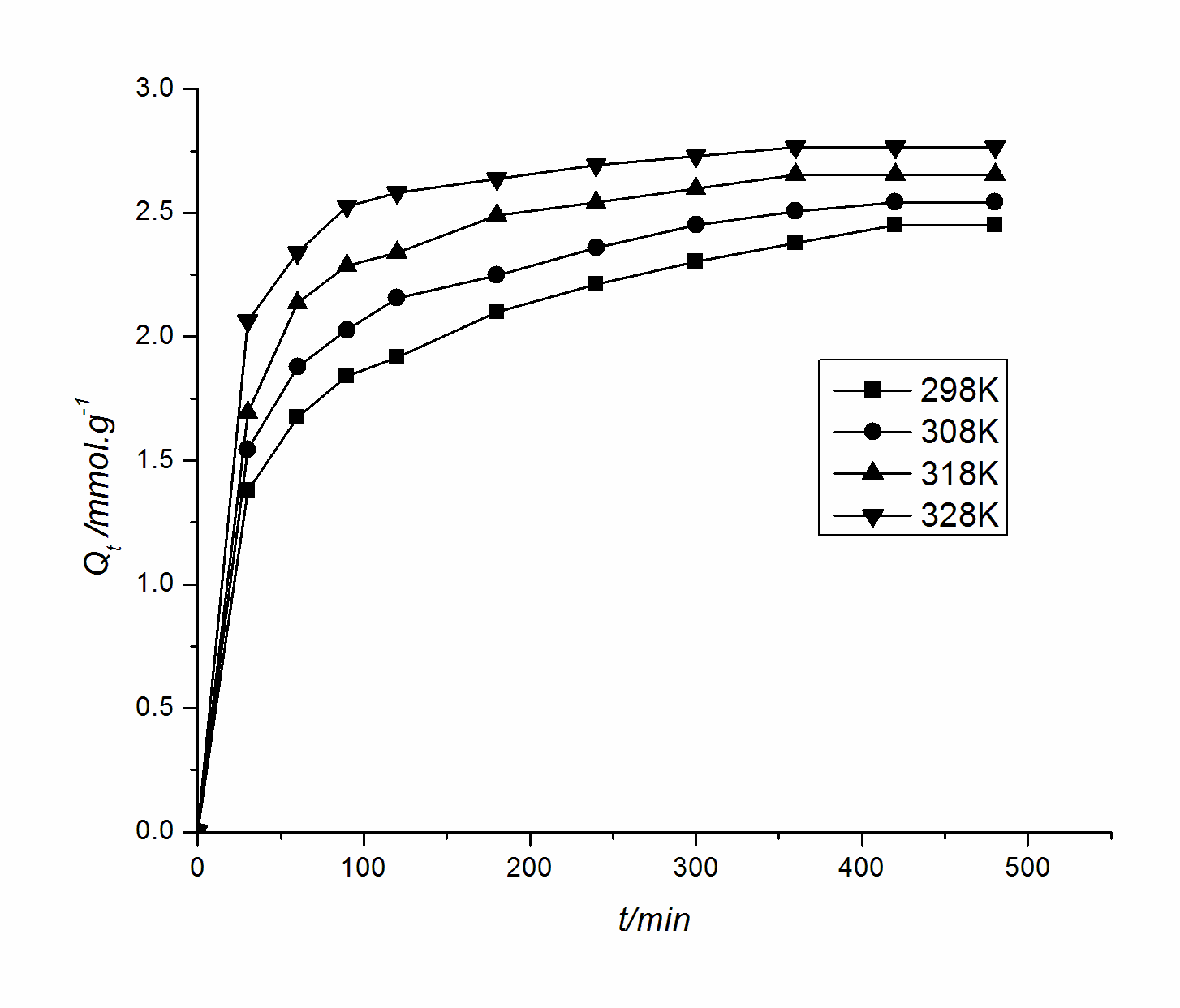
Kinetic curve of Ni 2+ adsorbed on aluminum oxide composites Figure 2 Pseudo-second order kinetics curve The pseudo-second order kinetic model is based on the adsorption rate determined by the square of the unoccupied adsorption gaps on the adsorbent surface. based on the assumption of [5] , the formula is : dQ T / dt = K (Q EQ -Q is T ) 2 (2) of formula (2) integral , and using the boundary conditions are obtained : T / Q T = T / Q eq +1/kQ 2 eq (3) where Q t is the adsorption amount when the adsorption time is t (min) (mmol × g -1 ); Q eq is the equilibrium adsorption amount (mmol × g -1 ); k Is the apparent adsorption rate constant (g × mmol -1 × min -1). You might be interested in reading the following post with recent chemistry news.

Adsorption meet pseudo second dynamics model, t / Q t and t is linear. Pseudo-second-order kinetic equations are fitted to the data in Figure 1 to obtain slopes and intercepts for the curves (see Figure 2), and to obtain Q eq and k values , respectively (see Table 1).
Table 1 Adsorption Kinetic Parameters at Different Temperatures
|
T/K |
Q eq /(mmol× g -1) |
k/(g× mmol -1× min -1 ) |
R 2 |
|
298 |
2.6312 |
0.00992 |
0.99813 |
|
308 |
2.6917 |
0.01301 |
0.99926 |
|
318 |
2.7729 |
0.01831 |
0.99984 |
|
328 |
2.8418 |
0.02876 |
0.99993 |
In lnk of 1 / T plot is a straight line, the correlation coefficient R & lt 2 = 0.9798. According to the Arrhenius equation, the apparent activation energy of adsorption is E a = 28.62 kJ × mol -1 .
 2.2 Complex combined material of Ni 2+ adsorption thermodynamics of
2.2 Complex combined material of Ni 2+ adsorption thermodynamics of
FIG. 3 shows that as of Ni 2+ increase in the initial concentration of the composite material of Ni 2+ adsorption capacity also increases, and then slowly change, tends to a maximum value . This is due to the limited number of adsorption sites when the dosing amount of the composite material is constant. When the initial Ni 2+ concentration is low, it can provide sufficient active sites for adsorption, and thus the adsorption amount of the composite material increases [ 6] . However, when the initial Ni 2+ concentration is high, the active sites are relatively insufficient, resulting in saturation of the adsorption sites. Therefore, when the Ni 2+ concentration reaches 0.25 mol × L -1 or more, the amount of Ni 2+ adsorbed by the composite is almost the same. Tend to a certain value.
The Langmuir adsorption model satisfies: C eq /Q eq =C eq /Q m +1/KL Q m (. 4)
where Q m is the saturated adsorption capacity ( mmol × G -1 ); C EQ and Q EQ are metal ions in solution equilibrium concentration ( mmol × L -1 ) and the adsorption capacity ( mmol × G – 1 ); K L is the equilibrium constant ( L × mmol -1 ).
The experimental datawas fittedaccording to the Langmuir adsorption model to obtain Figure 4 . Itcan be seenfrom Fig. 4 thatthe adsorptionof Ni 2+ by thecomposite material isin good agreementwith the Langmuir equation and is monolayer adsorption. By table 2It can be found that as the temperature increases, the saturated adsorption capacity and Langmuir equilibrium constant of the composite material also increase. Therefore, it can be seen that the adsorption belongs to the endothermic process, and the increase of the temperature favors the adsorption.
Table 2 Langmuiradsorption isotherm equations and parameters
| T/K | Langmuir equation | Q m /(mmol × g -1) | K L /(L × mmol -1 ) | R 2 |
| 298 | C eq/ Q eq =0.37413C eq+2.47580 | 2.673 | 0.1511 | 0.99986 |
| 308 | C eq /Q eq =0.36360C eq+1.82621 | 2.750 | 0.1991 | 0.99995 |
| 318 | C eq /Q eq =0.35394C eq+1.49414 | 2.825 | 0.2369 | 0.99996 |
| 328 | C eq /Q eq =0.34313C eq+1.25473 | 2.914 | 0.2735 | 0.99998 |
* Experiment of Ni 2+ concentration of L × 0.08 mol -1 – 0 .4mol × L -1 , adsorption time is 420min.
The relationship of K L with temperature T is given by Van’t Hoff equation [7] :
lnK L = – D H q /RT+ D S q /R (5)
where D H q (J × mol -1 ) and D S q (J × mol -1 × K -1 ) is the enthalpy change and the entropy change of the adsorption process, respectively; R is the ideal gas constant; T ( K ) is the temperature. Ln K L -1/ T is linear (R 2 = 0.9840 ), the slope and the intercept are equal to – D H q /R and D S q /R , respectively, from which D H q = 15.93 kJ× mol -1 , D S q =95.43J× mol -1× K -1 . The calculated D G q at each temperature is shown in Table 3 .
Table 3 D G q adsorption of Ni 2+ by chitosan – aluminum oxide composites at different temperatures
| T/ K | 298 | 308 | 318 | 328 |
| D G q /(kJ× mol -1 ) | -12.51 | -13.46 | -14.41 | -15.37 |
Gibbs free energy change at a temperature of 4 of the D G Q are less than zero, and the higher the temperature , D G Q the small, discloses composites of Ni 2+ adsorption is a spontaneous process, and the higher temperature The greater the degree of spontaneousness. D H q >0 indicates that the adsorption is an endothermic process. When the temperature rises, the energy of the reaction system increases. At the same time, the Gibbs free energy decreases, which is favorable for the adsorption of Ni 2+, so the adsorption amount increases.
3 Conclusion
Chitosan – aluminum oxide composites adsorbed Ni 2+ in aqueous solution in accordance with pseudo-secondary adsorption kinetics; the apparent activation energy of adsorption E a = 28.62 kJ × mol -1 . The adsorption of ions by chitosan – aluminum oxide composites conformed to the Langmuir adsorption isotherm in the studied Ni 2+ concentration range , and the adsorption thermodynamic parameters were obtained.
REFERENCES
[1] Jiang Tingda . Chitosan . Beijing: Chemical Industry Press, 2001.
[2] Xie GY, Du C Q. Ion Exchange and Adsorption (Lizi Jiaohuan Yu Xifu), 2009, 25(3): 200 -207.
[3] Dang MY, Zhang YA, Wang P, et al. Journal of Chemical Industry and Engineering (Huagong Xuebao), 2007, 58(5): 1325-1330.
[4] Huang YR, Li ZJ, Wang HF, et al. Applied Chemical Industry (Yingyong Huagong), 2009, 38(8):1093-1097.
[5] Sun XL, Zeng QX, Feng C G. Acta Phys.-Chim.Sin. (Wuli Huaxue Xuebao) ,
2009, 25(10): 1951-1957. [6] Chen YN, Ding Y C. Journal of Anhui Agri. Sci. (Anhui Nongye Kexue), 2009, 37(7): 3190-3192.
[7] Atia A., Donia AM, El-Boraey HA et al. Sep. Purif. Technol., 2006, 48(3): 281-287.
